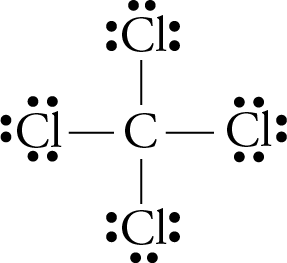
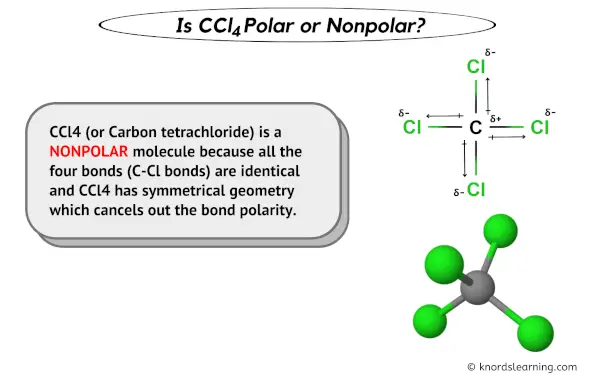
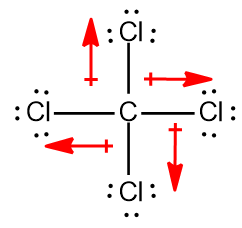
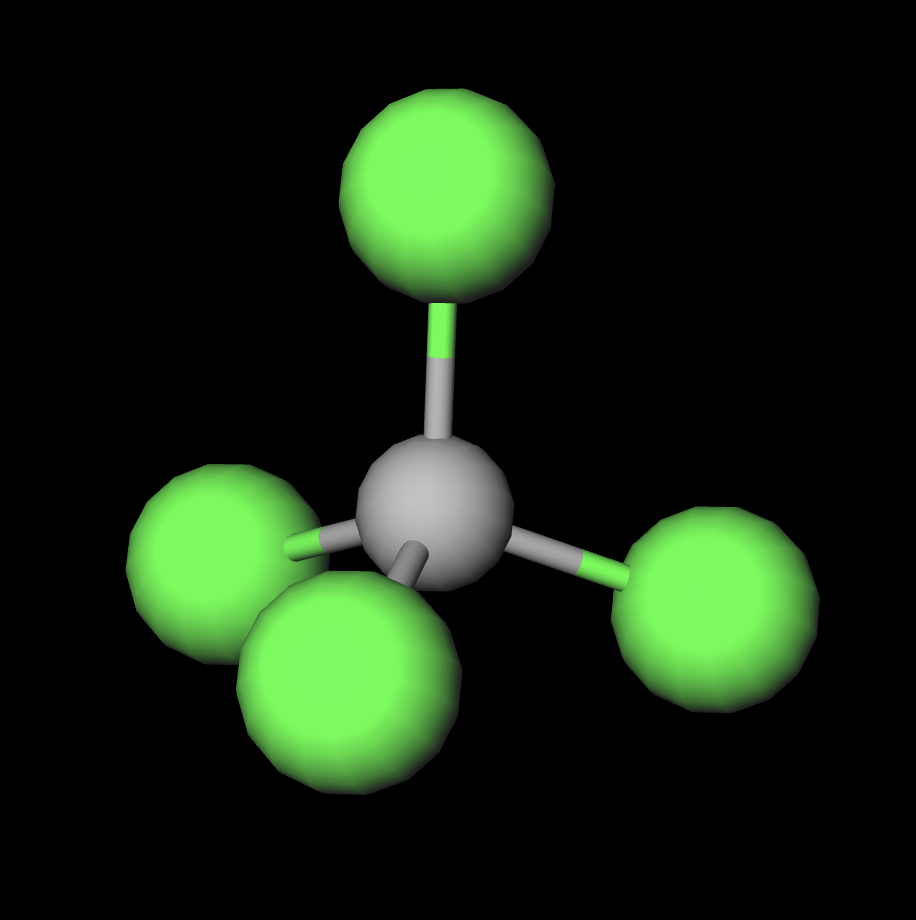
The carbon tetrachloride molecule (CCl4) is: A) a nonpolar molecule with polar bonds B) a nonpolar molecule with nonpolar bonds C) a polar molecule with polar bonds D) a polar molecule with

SOLVED: Explain how it is possible for CCl4 to have polar bonds but be a non-polar molecule. A diagram may be helpful in your answer, but it must be explained. The bonds
![DocMD #LENI2022 ✊🏼🇵🇭 💗 on Twitter: "[Chemistry] What is the polarity and shape of Carbon tetrachloride, CCl4? -Last question-" / Twitter DocMD #LENI2022 ✊🏼🇵🇭 💗 on Twitter: "[Chemistry] What is the polarity and shape of Carbon tetrachloride, CCl4? -Last question-" / Twitter](https://pbs.twimg.com/media/D0gM7FuV4AA1fyM.jpg)